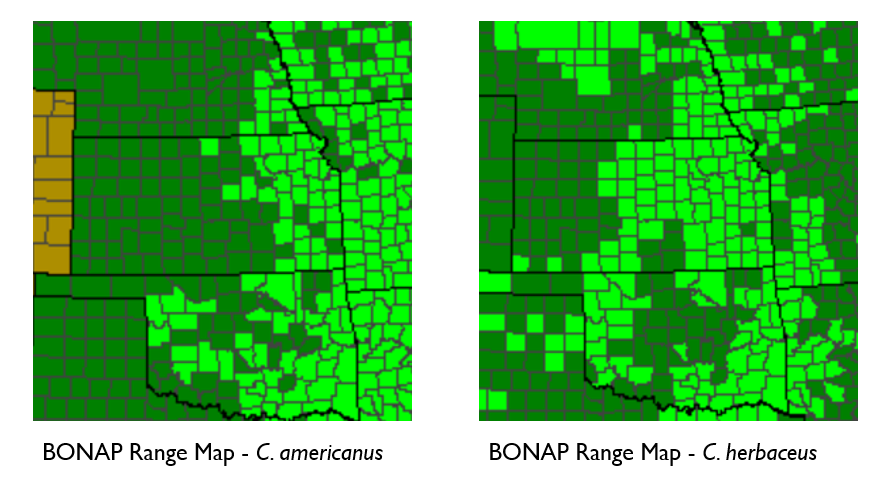
This February I had an opportunity to visit the monarch butterfly overwintering grounds in the Sierra Madres mountains of Mexico. It was AMAZING! Yes, really, it was all-caps level cool. The trip was an absolute dream that I will surely be retelling for years to come. Starting now! Read on for info on the monarchs, their incredible migration, and my experience visiting their wintering grounds.
Short Flight for Me, Long Flight for Them
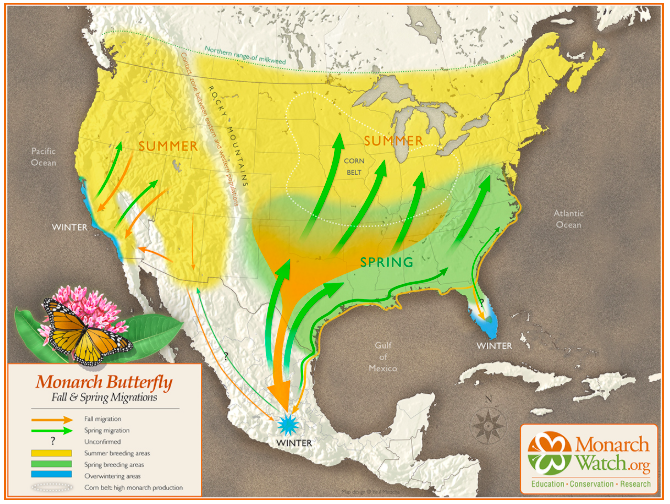
I flew into Mexico City in late February, which is the perfect time of year to visit the monarchs. DFT to MEX is a quick three hour flight. But as I was floating up above the clouds, cozy in my seat with a beverage and a snack, I thought of how much tougher the monarchs have it! Starting in the northern US and Canada, they depart in late summer and fly for 4-8 weeks to reach Mexico by late October. Seemingly so fragile, with tiny bodies and impossibly thin wings, yet they endure so much – rain, wind, hailstorms, heatwaves, miles and miles of monocrop agriculture without a nectar plant in sight to fill their bellies! It seems like slim odds that any survive the trip, but somehow they do.
High Altitude Hike
Once in Mexico, they gather in a particular part of the Sierra Madres mountain range. In total, their overwintering area covered 7.24 acres this winter, which is a 64% increase over last winter. This is great news! Their favorite spots are located within the state of Mexico and the state of Michoacán, in high mountain elevations between 9,000 and 11,000 feet. They are choosy creatures, preferring to gather on in areas with Oyamel fir trees (Abies religiosa). For the visitor, this means traveling a few hours from Mexico city by car, then an hour on horseback up the mountain, then another 45 minutes to an hour on foot to reach their roosts. Local guides and sanctuary employees keep tabs on the butterflies’ location, and change the footpaths as needed, so the hike may be longer or shorter depending on where they are at when you visit.

Wonderland
The travel time and hike, no matter how long, is worth it. As you get closer, you might see a single monarch flitting across your path, and then another, and another. Suddenly, you’re in a cloud of them! My visit day was warm and sunny, so the monarchs were very active. They flew overhead in every direction, bordering on a swarm. I hardly knew where I was walking, as I was looking up instead of down. The narrow dirt paths are roped in these areas, probably to keep us from falling to our deaths from not watching where we step. But also to keep visitors from getting too close to the roosts, where thousands (maybe millions?) of butterflies hang off the tree branches in fluttering clusters.
Photos below taken by another traveler in my group, Jennifer Elise at Journey with Jennifer LLC.



The Sound of Monarchs
Sanctuary guides and locals may journey up the mountain with visitor groups simply to enforce the cardinal rule: Shhhh! Remaining quiet, so as not to disturb or startle the butterflies, is important. And it also enhances the sensory experience — the quiet of the forest is punctuated by bird call, rustling leaves, and wind through pines. And the most magical of all sounds? Butterfly wings! Thousands of them together beating the air makes a faint sound like moving water.
Needless to say, I was transfixed. I could have stayed there for hours, watching them fly and feed and mate and fight. Insects are more complex than we give them credit for, and this awe-inspiring phenomenon is a reminder of that. All insects, from charismatic butterflies to the humble cricket, are fascinating and essential parts of our ecosystem. Stay tuned for a follow up of this blog, detailing the flowers of the Oyamel understory (the blooms these monarchs feed on in spring, their final meal before heading north again!) and tips for some native Kansas analogs you can plant for them to sip on once they reach us here.