The Essential Foundation of the Butterfly Garden
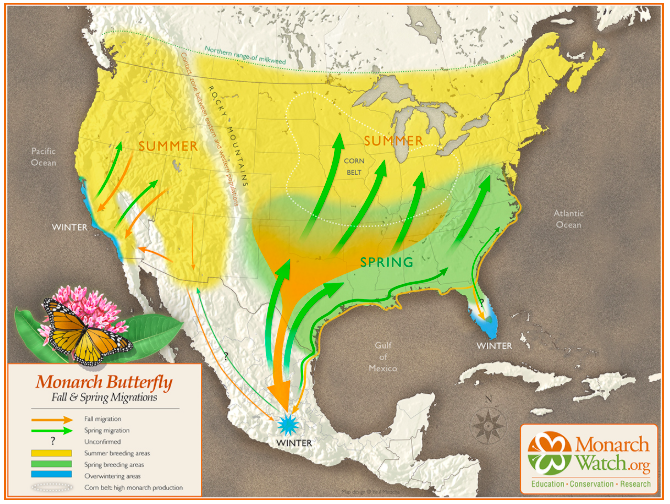
Native milkweeds (genus Asclepias) are far more than just roadside weeds; they are the biological foundation of our resilient North American landscape. As the sole host plant for Monarch butterfly larvae, milkweeds provide essential chemical compounds that protect these iconic insects from predators. By integrating native species into your garden, you aren’t just adding greenery—you are participating in a larger conservation effort to stabilize declining pollinator populations.

Selecting the Right Species for Your Soil
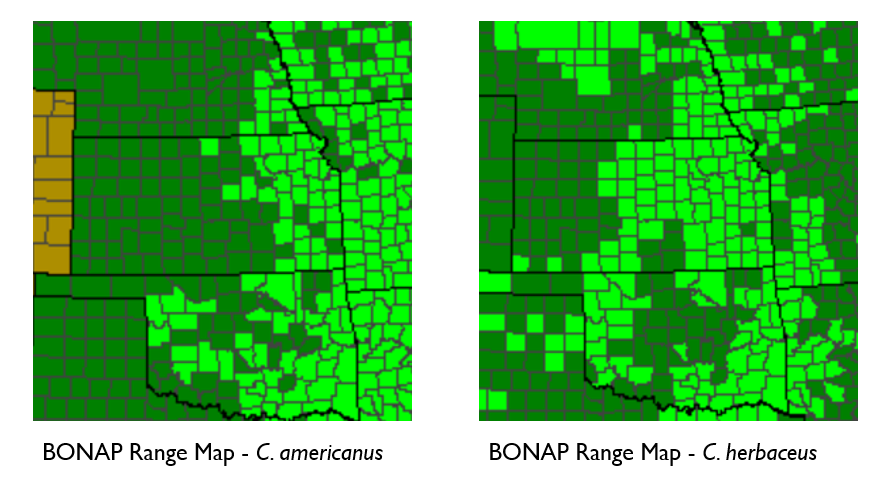
One of the most common misconceptions is that all milkweeds require the same environment. To ensure success, you must match the plant to your specific site conditions. Swamp Milkweed (Asclepias incarnata) thrives in heavy clay and moist soils, making it perfect for rain gardens or along a pond edge like we have at the Arboretum, while Butterfly Weed (Asclepias tuberosa) demands sharp drainage and sunny, dry spots. Choosing a species indigenous to your specific ecoregion ensures the plant will thrive with minimal supplemental watering once established.

Aesthetic Appeal Beyond Utility
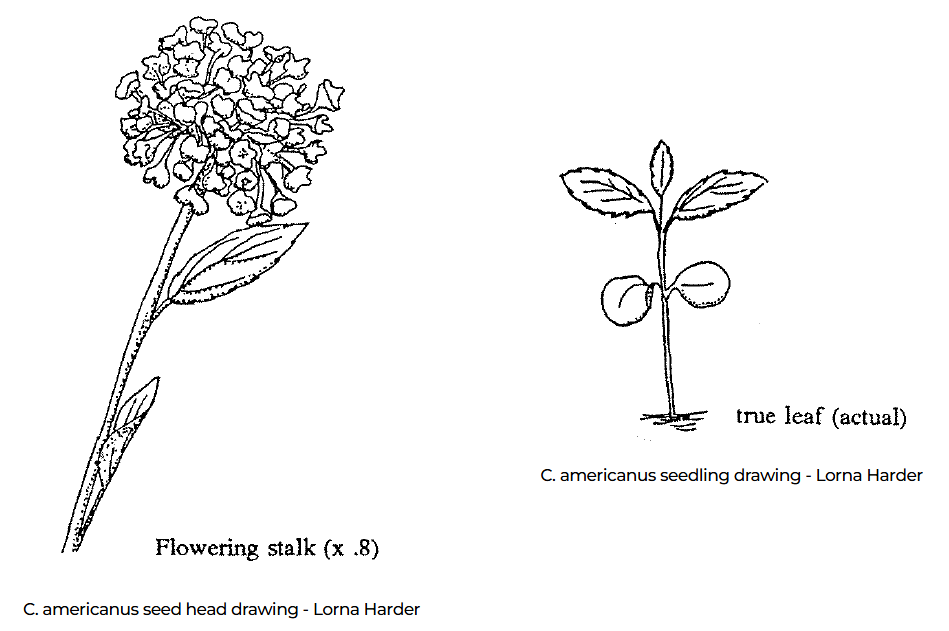
Beyond their ecological impact, native milkweeds offer striking ornamental value that rivals traditional nursery bedding plants. The vibrant, tangerine clusters of Butterfly Weed provide a rare “true orange” to the summer border, while the architectural, spherical blooms of Common Milkweed (Asclepias syriaca) fill the evening air with a heavy, honey-like fragrance. Other spreading but valuable milkweeds are Sullivant’s Milkweed (Asclepias sullivantii) and Showy Milkweed (Asclepias speciosa). Each milkweed has unique seed pods that also provide late-season visual interest, eventually opening to release silken “floss” that catches the autumn light.

Managing Growth and Expectations
Integrating milkweed into a formal landscape requires a bit of strategic planning. Some species, like Common Milkweed, Sullivant’s Milkweed, and Showy Milkweed spread aggressively via underground rhizomes and are best suited for meadow-style plantings or large back-borders. Plant these spreading milkweeds with other pushy natives like beebalm, spreading goldenrods, gray headed coneflower and Brown eyed susans and let them fight it out. For smaller urban plots, “clump-forming” varieties like Whorled Milkweed (Asclepias verticillata), Green Antelopehorn Milkweed (Asclepias viridis), and Butterfly Milkweed offer a more refined, airy texture that won’t overtake neighboring perennials. Deadheading spent blooms can prevent unwanted self-seeding, though leaving a few pods is vital for the next generation of plants and silk-gathering birds.

A Year-Round Wildlife Hub
The value of milkweed in the landscape extends well beyond the Monarch. The high-quality nectar produced by Asclepias flowers supports a diverse “who’s who” of beneficial insects, including honeybees, specialized milkweed bees, and predatory wasps that provide natural pest control. Even in winter, the sturdy stalks can provide nesting habitat for native bees. By choosing native milkweeds over exotic alternatives like Tropical Milkweed—which can harbor parasites—you create a safe, synchronized, and spectacular sanctuary right in your own backyard.